COVID-19 presents a unique opportunity to accelerate and expand national policies on differentiated service delivery (DSD) for people living with HIV (PLHIV). DSD is “a responsive, client-centred approach that simplifies and adapts HIV services across the cascade to better serve individual needs and reduce unnecessary burdens on the health system.” Under the umbrella of DSD are a range of strategies designed to make it more convenient and comfortable for individuals to access HIV services.
Multi-month dispensing is one such strategy.* Since 2016, the WHO has recommended that PLHIV who are stable on antiretroviral therapy (ART) be given 3- or 6-month medication refills in order to minimize pharmacy and health clinic visits. Now, given COVID-19, the WHO recommends that all PLHIV on ART have at least 30 days of medication with them, and ideally a 3-6 month supply.
As people all over the world are being asked to stay-at-home, UNAIDS, WHO, and others are calling on countries to expand DSD policies, including MMD. Over the last few weeks, the HIV Policy Lab has compiled national policies on multi-month supplies of HIV medication for 126 countries. This crucial data will help target the efforts of UNAIDS, GNPPlus, policy advocates, and others in the HIV-community urgently calling on countries to adopt multi-month dispensing and ensure sufficient access to ARTs during COVID-19.
Our Global Health Policy & Politics team gathered national policy documents (national HIV treatment guidelines, DSD guidelines, national HIV strategic plants, and pharmaceutical guidelines) to ascertain whether national policies allow for at least 3-month and at least 6-month dispensing of HIV medication for stable patients. We also looked at whether national policies clearly articulate who is considered stable on ART, and therefore eligible for multi-month dispensing. If national policy documents were not available, we supplemented with information from secondary sources including PEPFAR, NCPI, and WHO.
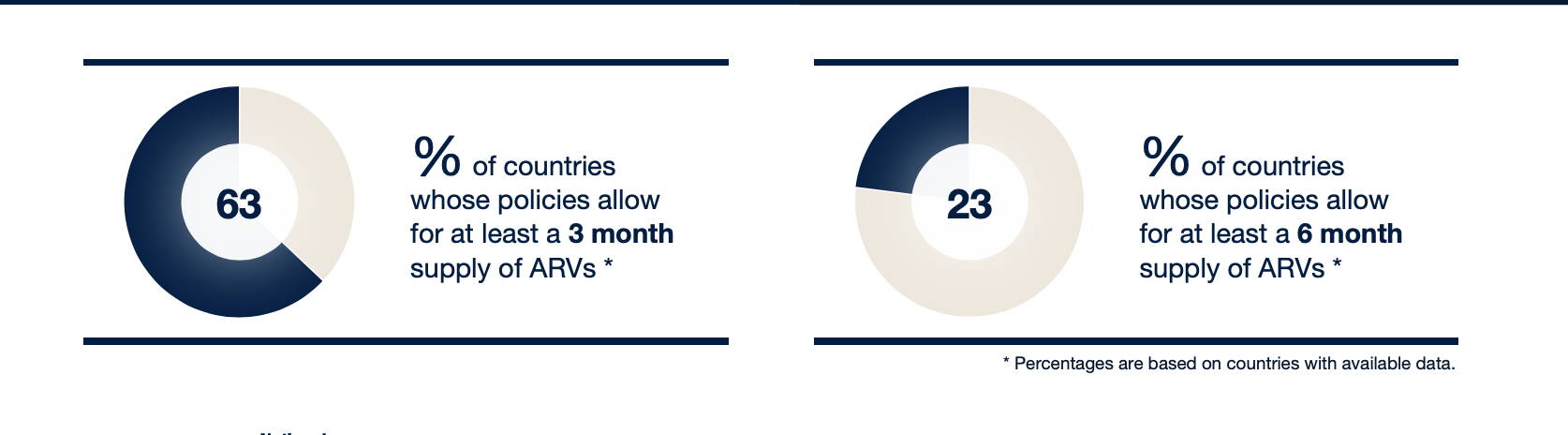
Of the countries with available data, 63% of country policies allow for at least a 3-month supply of ARVs, but only 23% allow for at least a 6-month supply of ARVs. Results varied by region, most notably East and Southern Africa. With the exception of Madagascar, Comoros, and Eritrea, all East and Southern African countries have policies allowing for at least 3-month dispensing. This is not surprising given the significant amount of HIV-focused aid targeted in this region over the past few decades. Similarly, PEPFAR countries** have at least 3-month dispensing, since this is now a requirement for PEPFAR countries to continue receiving funding.
However, donors promoting policies can only go so far. For example, South Africa has the largest number of PLHIV of any country and is a global leader in HIV policy, but its current policy provides only 2-month dispensing. In response to COVID-19, South Africa’s National Department of Health issued guidelines that call for 3-month dispensing in principle, but note that “[a]t this stage, South Africa is not in a position to move forward with multi-month dispensing for ART or PrEP for periods longer than 2 months, with the exception of 3 month dispensing [for one regimen] and the 6 month pilots, due to instability of stock supply globally.” In short, expanding HIV programs requires capacity as well as policy change. South Africa presents a perfect example of greater challenges in accelerating ART access such as supply chain capacity, stockouts, and other procurement challenges.
Of the high-income countries for which data was available, only about 39% of policies allow for 3-month dispensing and only about 19% of policies allow for 6-month dispensing. This seemingly significant lack of progressive policies represents yet another complication in HIV access – in most high-income countries prescription length primarily depends on insurance and pharmaceutical regulations rather than national guidelines, most of which were not constructed with HIV in mind.
Several countries have already made DSD policy changes given COVID-19, such as expanding the group of patients eligible for multi-month dispensing from only stable patients to all patients on ART. However, national policies on multi-month dispensing remain largely inadequate. COVID-19 presents an urgent need to assess the status of MMD policies globally as well as a unique opportunity to track and analyze both short and long-term policy change.
For full results on national policies for multi-month supplies of HIV medication go to https://www.hivpolicylab.org/ and download our policy tracking brief.
For more information on HIV and COVID-19 please visit https://www.unaids.org/en/covid19.
* Other DSD strategies include community-based antiretroviral therapy (ART) delivery, peer-led adherence support groups, and multi-month dispensing (MMD). For more information on DSD, please visit http://www.differentiatedservicedelivery.org/.
**Angola, Botswana, Burundi, Cambodia, Cameroon, Côte d’Ivoire, DRC, Dominican Republic, Eswatini, Ethiopia, Ghana, Haiti, Indonesia, Kenya, Laos, Lesotho, Malawi, Mozambique, Namibia, Nigeria, Rwanda, South Africa, South Sudan, Tanzania, Thailand, Uganda, Ukraine, Vietnam, Zambia, & Zimbabwe.



