Tomorrow is Commissioner Gottlieb’s last day on the job at the U.S. Food and Drug Administration (FDA), and he’s been treating these last few days much like he has his entire two-year tenure: with carefully thought out and coordinated communication about both in-process and potential future FDA actions. While the agency has been hard at work in essentially all of their regulated product areas, the recent progress on tobacco and nicotine products regulation really has been quite extraordinary. I say this as someone with strong tobacco control advocacy roots who also spent a year at the FDA’s Center for Tobacco Products (CTP) begrudgingly shifting her frame from harnessing the powerful new tools to regulate tobacco products in ways that would improve the public health to simply better understanding the many limitations—both perceived and real—to achieving that same goal.
Given that tobacco still kills more people each year than alcohol, illegal drugs, AIDS, murders, suicides, and car accidents combined, I’m not saying I agree with all of FDA’s actions or that the pace of forward progress is satisfactory, but I also think giving credit where credit is due is important. I also know my former CTP colleagues have been working diligently for many years on the proposals we see moving forward. Just as critics outside the FDA debate about how and when the agency should be doing things differently, so too do those on the inside as they navigate the realities of putting together each of their pieces of the regulatory puzzle. Their steadfast efforts deserve thanks and recognition, especially now that they’re finally making their way into the public eye.
For more concrete perspective on just how extraordinary FDA’s forward progress has been, let’s revisit the timeline for one of the agency’s most seminal tobacco products rulemakings. I was lucky enough to be part of the CTP team that drafted the “deeming rule,” which allowed FDA to finally reach all tobacco products on the market. I was late to the party, joining only in the final rule stage, but the effort—from planning and proposed rule development to final rule publication—took almost seven years in total. Putting aside whether it should’ve taken that long, the reality is it that it did. While I want to see binding rules of engagement placed on a historically bad-acting industry just as much as many of my fellow public health colleagues, I also respect the current approach the agency is taking. Not only are they in the thick of deeming implementation (and litigation), but they’ve also been especially nimble under an Administration that promised deregulation.
Under Commissioner Gottlieb and CTP Director Zeller leadership, the FDA has operated under a bold, comprehensive framework for tobacco and nicotine regulation. First announced in July 2017, the framework clearly acknowledges a continuum of risk among products delivering nicotine and identifies a number of regulatory actions along that continuum, including for nicotine products regulated under FDA’s medical product authorities. In April 2018, no longer able to ignore the popularity of e-cigarette use among young people, the agency also announced new enforcement actions and a youth prevention plan “to stop youth use of, and access to, JUUL and other e-cigarettes.” What has ensued ever since is an evidence-based recalibrating of the risks and benefits e-cigarettes pose to overall public health.
And FDA has been considering all kinds of evidence about e-cigarettes. From misleading labeling and/or advertising to other industry marketing practices to national youth survey data from 2017 and 2018, the agency is also actively responding to what they’re finding. FDA has:
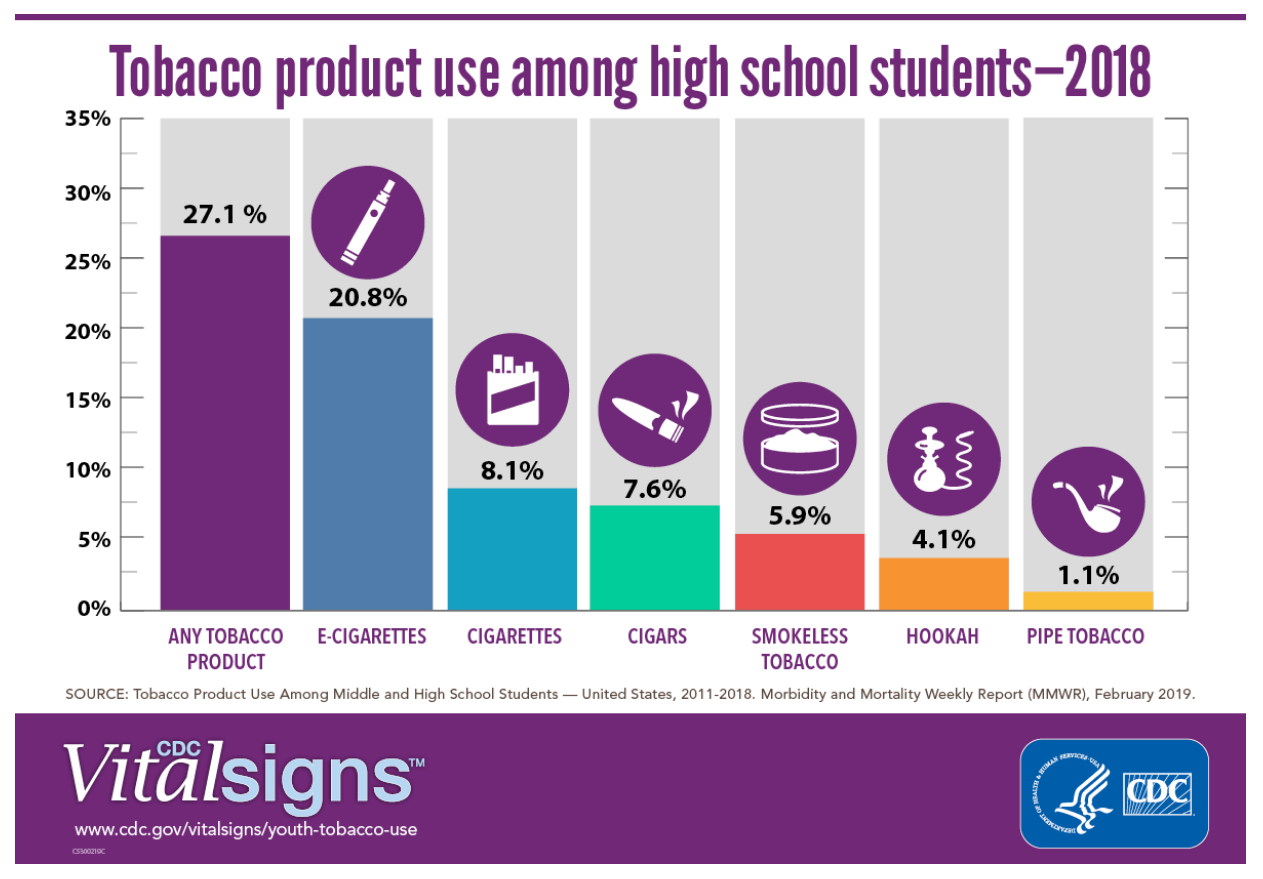
- Ramped up enforcement and education efforts targeting youth use of e-cigarettes,
- Begun to tread enforcement paths at the intersection of their tobacco and medical product authorities and that clarify tobacco product premarket review requirements (see here and more here),
- Applied public pressure to e-cigarette manufacturers to “step up” and explain seemingly inconsistent representations made to the agency by two companies (see Letters to JUUL Labs, Inc. and Altria Group Inc.),
- Proposed new flavored e-cigarette enforcement policies, and
- Warned about potential seizure risk associated with the use of e-cigarettes.
This isn’t even a complete list of their efforts specific to e-cigarettes. Nor does it mention important FDA signals and developments to move the needle for conventional cigarettes, other tobacco products, and medicinal products to help both youth and adults addicted to nicotine to quit. Peruse the agency’s major public-facing actions.
Based on the almost-seven-year deeming rulemaking benchmark I began with, I’m not surprised we haven’t seen more rulemaking initiated. And the substantial equivalence proposed rule that was just recently published is an important foundational rulemaking to clarify a number of premarket review issues, including what types of changes result in a “new tobacco product” and whether definitions for “test marketing” and “commercially marketed” are warranted. As with all FDA actions (or inaction in some cases), it will surely prompt FDA critics to continue their advocacy on all sides of the issue. I don’t expect, or want, my reflections to quiet the advocates, and I’m not urging complacency. I simply think it’s worth taking a moment to appreciate the progress that has been made.
* Image courtesy of CDC Vital Signs



